The natural process of turning metals into undesirable compounds when they come into contact with corrosive fluids like water or air is known as corrosion.
Corrosive environments change metals into their corresponding oxides, sulfides, and hydroxides (for example, iron rusts), which lead to damage and metal disintegration that begins with the exposed area of the metal and spreads to the entire metal.
Option 1: Select from a range of popular corrosion-resistant metals.
Stainless Steel
A family of roughly 200 steel alloys with strong heat and corrosion resistance is known as stainless steel. The carbon content ranges from 0.03% to 1.2%, but its high chromium content makes it special. About 10.5% of stainless steel is made of chromium, which forms a passive oxidation layer that shields the metal from corrosion.
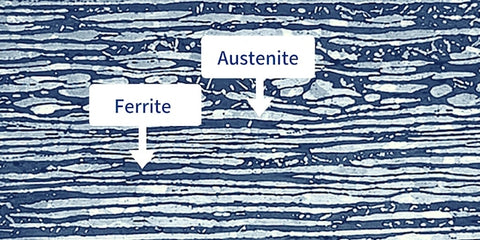
Stainless steel can be divided into three major groups according to its microstructure:
- Austenitic Stainless Steel: Austenitic stainless steel has 8–12% nickel and at least 18% chromium. Nitrogen, carbon, and a variety of other elements are also present. High corrosion resistance is provided by chromium, while nitrogen stiffens the material. The 300-series stainless steels are an assortment of alloys, and two typical grades are stainless steel 304 and stainless steel 316, the latter of which is the most corrosion-resistant metal.
- Martensitic Stainless Steel: Martensitic stainless steel typically includes 11.5–13% chromium, 0.15–0.15% carbon, 0.1% manganese, molybdenum, and sulfur or selenium. The range is known as the 400-series, and grade 420A is the most popular. The alloy is strengthened by the carbon, however it is less corrosion resistant than the 300 series due to the low chromium level.
- Ferritic Stainless Steel: Also known as a straight chromium non-hardenable class of stainless alloys, ferritic stainless steel has a carbon content that is less than 20% and a chromium content that ranges from 10.5% to 30%. Cold rolling can somewhat harden these steels, however heat treatment cannot. While the 430A is the common grade, these are also in the 400-series.
Duplex Stainless Steel
The name "duplex steel alloy" implies that it is composed of two distinct phases. In order to create the most advanced corrosion-resistant metal, duplex steels combine the greatest qualities of the ferritic and austenitic phases. S32750, which contains 25% chromium, 7% nickel, and 4% molybdenum, and 2205, which contains 22% chromium, 5% nickel, and 3% molybdenum, are typical grades. They are employed in numerous difficult operations.
Superalloys
Superalloys are high-performance metal alloys that provide superior mechanical qualities and corrosion resistance at high temperatures. Because of this, certain types of corrosion-resistant metals are frequently employed in applications that require both high performance and corrosion resistance (like aerospace and medical).
Superalloys can be distinguished by their primary matrix component:
- Nickel Superalloy: Due to their low coefficient of thermal expansion, nickel-based superalloys not only exhibit good corrosion resistance but also great strength and heat resistance. These materials are unique because of their outstanding machinability and form memory. One material that can be 3D printed is inconel 718.
- Cobalt Superalloy: Compared to their nickel- or iron-based equivalents, cobalt-based superalloys have greater melting points. In comparison to nickel- or iron-based alloys, they also provide exceptional hot corrosion resistance. Compared to alloys based on nickel, cobalt-based superalloys are very weldable. Powders of cobalt chrome are 3D-printable.
- Iron Superalloys: are very strong and very resistant to oxidation, creep, wear, and corrosion at standard room temperature. They cost a lot less than the first two options.
Aluminium
Due to its strong affinity for oxygen, aluminum forms a passive oxide layer that protects it against corrosion. The majority of aluminum grades are resistant to chemicals, but the strongest grades are 1xxx, 3xxx, and 5xxx. They are resilient in the generally corrosive pH range of 4.5 to 8.5. An excellent method for giving regular aluminum an anti-corrosion covering is anodizing.
- 1xxx: The best corrosion resistance in typical applications can be found in the 1xxx, which is extremely pure (around 99%).
- 3xxx: (Al-Mn alloys with a maximum Mn content of 1.25%), manganese serves as the primary alloying element. As a result, it has a slightly lower resistance to corrosion than 1xxx aluminum. However, manganese gives the alloy its ductile properties. Al-Si1Mg aluminum, for example, resists corrosion cracking at high stress.
- 5xxx: This sort of aluminum's primary alloying component, magnesium, provides corrosion resistance practically on par with aluminum of the 3xxx grade. It has good corrosion resistance and a high rate of work hardening. The surface polish is brilliant as well.

Copper Alloys
High thermal conductivity, resistance to corrosion, exceptional hardness, and high-temperature strength are all characteristics of copper alloys. In order to increase the corrosion resistance of other metal alloys, copper is also added to them, which has an excellent corrosion resistance on its own. The copper alloy family's most popular corrosion-resistant metals are:
- Bronze: One of the first materials ever discovered is bronze. 12% tin and 88% copper make up modern bronze. Nickel, manganese, aluminum, silicon, zinc, or arsenic may also be present in bronze. The alloy's corrosion resistance is improved by the trace amounts of silicon added.
- Brass: Brasses are copper and zinc alloys. Tin may also be included in trace levels to add additional corrosion resistance, although zinc may have the opposite effect. Brasses often have a lower melting point than bronze or even pure copper, as well as good tensile strength and high corrosion resistance.

source: brass vs gold (https://learnaboutgold.com)
Titanium
Because it resists corrosion and has a strong strength to weight ratio, titanium is a very durable metal that is frequently utilized in technical applications. Despite being 40% lighter than steel, it is just as strong.
Titanium creates passive oxides just like other corrosion-resistant metal compounds, without which it corrodes right away. The ability of titanium to withstand chlorine is a special characteristic. The alloy titanium 3.7164 (Titan Grade 5) is a superb illustration of a material that resists corrosion.
Option 2: Enhance parts' corrosion resistance using post-processing treatments
Thanks to a variety of surface finishing options, the corrosion resistance of CNC machined, sheet metal, and 3D printed items can also be improved:
Anodizing: When a metal is machined (mostly aluminum parts), the surface is hardened by an oxidation process to make it more resistant to wear.
Processing of spray paint: Spray paint protects objects from rust, including pipelines, electrical enclosures, and other items used every day.
Electroplating: To prevent corrosion, another non-corrosive metal is electroplated over the component's surface. To extend the lifespan of standard metals, the corrosion-resistant metals listed below are occasionally coated on them.
Surface polishing: To remove the sharp burrs left behind from the machining process, surface burr processing is done to CNC-machined items. It is suggested that the sharp edge of the corner be polished into a smooth face so that it won't hurt people when they use it.

spray painting a stainless steel surface